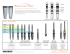Preliminary Evaluation of C1 Implants
Transcription
Preliminary Evaluation of C1 Implants
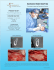
30 News No. 30 June 2012 Preliminary Evaluation of C1 Implants ® Make it Simple 2 News 30, June 2012 Preliminary Evaluation of C1 Implants Jorge Arango ; 2 Leonardo Calvache ; 2 Miguel Ferrigno 1 Introduction Conclusions The Center of Oral Implantology at the Colombian School of Dentistry has evaluated a new implant system from MIS Technologies LTD, C1 Implant, are conical, self-tapping implants with three parallel micro rings in the coronal area. The surface treatment is sandblasting acid etched. Its conical connection provides platform switching as well as internal anti-rotation mechanism. The implants are color-coded. The conclusions of the initial evaluation of the surgical phase using twelve C1 implants ,in five implant cases are: ▪ The surgical kit and its design is non-complex, logical and easy to follow the correct sequence. We selected five patients who needed between one to four implants each. Four cases were at the mandible with type I and type II bone. One at the anterior maxilla with type III bone. Our surgeons, Dr. Calvache and Dr. Ferrigno used twelve implants; four 10mm long and four 11.5 mm long, in size 3.75. One 10 mm long and three 11.5 mm long, in size 4.2. None of them were immediately loaded. At the surgical phase, we evaluated the following items: drilling sequence, delivery to site, insertion, bone stability, cover screw or healing cap handling and an overall evaluation. ▪ The color code really helps to identify different platforms. ▪ The direction indicator tools facilitates the identification of the angle of the implant and determines the correct three dimensional position. ▪ The stoppers on the pilot drills help to determine the correct length. ▪ The conical root-shaped, the thread and the self-tapping design as well as the mild bone compression, enhance the implant's primary stability. ▪ The conical connection offers better seal and minimizes micro-movements and support preservation of bone. ▪ The micro rings and the dual thread design on the implant’s neck improve bone to implant contact at the crestal zone. This initial evaluation presents positive results. However, it is necessary to develop research studies to come to stronger conclusions based upon scientific evidence. ▪ The final disposable drill easily cuts through bone. ▪ The double packing system ensures sterilization and safety issues. ▪ The insertion system is very convenient and minimizes the number of tools. Patient Report Case 1 Name C.G Age 46 2 J.L 25 3 O.C 78 4 O.S 36 5 L.P 47 Implants Implants Diameter(mm) Length (mm) 46 4.2 11.5 47 4.2 11.5 34 41 46 47 34 3.75 3.75 3.75 3.75 3.75 10 10 10 10 11.5 44 4.2 10 45 4.2 10 47 3.75 11.5 22 3.75 11.5 12 3.75 11.5 UNICOC (Institucion universitaria colegio odontologo Colombiano) is a leading institute for Odontological studies in Colombia. It was established by Prof. Jorge Rene Arango Tamayo and received its recognition as an academic institute by the national ministry of education in 1975. It acts as an active member of the Colombian Association of dentistry faculties (ACFO) since 1981, and as a founding member of the Colombian association of private university institutions (ACIUP) since 1988. UNICOC 's vision is to become a world leader through extensive research activities, through broad scientific educational and training programs, through innovation, and through social responsibility. 1 Prosthodontist, Postgraduate Professor, Oral Implantology Center Colegio Odontologico, Institución Universitaria Colegios de Colombia UNICOC. 2 Oral and maxillofacial surgeon, Postgraduate Professor, Oral Implantology Center Colegio Odontologico, Institución Universitaria Colegios de Colombia UNICOC. 3 News 30, June 2012 CT Scan and X Rays Pre Surgical Post Surgical Case 1 Fig. 2 Case 2 Case 3 Case 4 Case 5 MIS’s Quality System complies with international quality control standards: ISO 13485:2003 - Quality Management System for Medical Devices, ISO 9001: 2008 – Quality Management System and CE Directive for Medical Devices 93/42/EEC. MIS’s products are cleared for marketing in the USA and CE approved. © MIS Corporation. All rights Reserved MC-N3012 Rev.1 ® MIS Implants Technologies Ltd. www.mis-implants.com
Similar documents
Preliminary Evaluation of C1 Implants
 UNICOC (Institucion universitaria colegio
odontologo Colombiano) is a leading institute
for Odontological studies in Colombia.
It was established by Prof. Jorge Rene Arango
Tamayo and received its ...
UNICOC (Institucion universitaria colegio
odontologo Colombiano) is a leading institute
for Odontological studies in Colombia.
It was established by Prof. Jorge Rene Arango
Tamayo and received its ...